Introduction: Elevated pro-inflammatory cytokines are a hallmark feature of myeloproliferative neoplasms (MPNs). The pro-inflammatory cytokine interleukin-8 (IL8) is increased in patients with myelofibrosis (MF) and correlates with adverse outcome, including overall survival. Previously, the Levine/Fang labs identified increased IL8 secretion from individual CD34+ stem cells in a subset of MF patients. The role of IL8 and its cognate receptors CXCR1/2 in MF pathogenesis has not been delineated.
Methods: Single-cell cytokine assays were performed on isolated CD34+ cells from 60 clinically annotated MPN patients (20 MF, 20 PV, 20 ET) using a previously described micro-chip platform (Kleppe et al, Can Disc 2013). 10 healthy donors (CD34+ cells from hip replacements) were used as controls. Integrated RNA-Seq and Assay for Transposase-Accessible Chromatin followed by next-generation sequencing (ATAC-Seq) was performed on CD34+ cells from MPN patients with and without expanded IL8 secreting clones for gene expression/chromatin accessibility analysis. To model the role of IL8-CXCR2 on fibrosis in vivo, the human MPLW515L transplant model (hMPLW515L) of MF was used. Specifically, wild-type (WT) murine bone marrow (Creneg-Cxcr2f/f; Cxcr2WT) or marrow lacking the CXCR2 receptor (VavCre-Cxcr2f/f; Cxcr2KO)were retrovirally infected with MSCV-hMPLW515L-IRES-GFP and transplanted into lethally irradiated WT recipient mice and monitored for disease. Blood counts, chimerism, and flow cytometry were assayed. Moribund mice were sacrificed and assayed for grade reticulin fibrosis and overall survival.
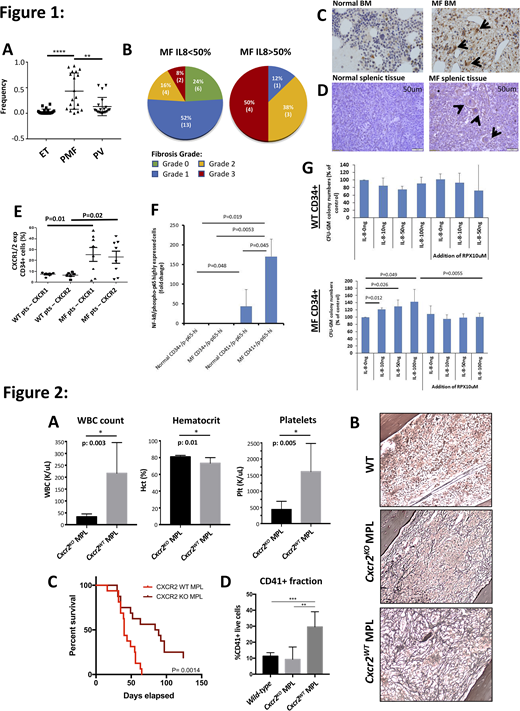
Results: Single-cell cytokine assays confirmed an increased proportion of IL8-secreting CD34+ cells in MF patients (40%) in comparison to other MPN sub-types (10% PV/0% ET) (Figure 1A). MF patients with expanded IL8 secreting clones (defined as >50% of total CD34+ cells) had increased leukocytosis (p<0.0001), larger spleen sizes (p=0.0004), greater prevalence of constitutional symptoms (p=0.0084), and higher-grade reticulin fibrosis in marrow (Figure 1B) in comparison to MF patients without prevalent IL8 clones. IHC confirmed increased IL8 expression in marrow biopsies from 8/15 MF patients in comparison to 0/4 normal controls (Figure 1C), and high IL8 expression was also observed in MF splenic megakaryocytes (MKs) as well as in splenic stromal/endothelial cells not seen in normal spleen (Figure 1D). Integrated RNA-Seq/ATAC-Seq analysis of IL8-high MF patients confirmed up-regulation of IL8-CXCR2 signaling and enrichment in pro-inflammatory pathways (i.e TNFa, NFkB, etc) by GSEA, as well as increased expression/accessibility of pro-inflammatory genes S100A8 and S100A9-previously implicated in fibrosis development. Flow analysis of IL8-high MF CD34+ cells revealed enhanced surface expression of CXCR2 and its analog CXCR1, such that MF was characterized by increased IL8 ligand and receptor expression (Figure 1E) and coincided with enhanced NFkB pathway activity (Figure 1F). Consistent with this, colony forming assays of cultured MF CD34+ cells revealed enhanced colony output when cultured with IL8 compared to WT CD34+ cells-an effect ameliorated by co-treatment with the CXCR1/2 antagonist Reparixin (Figure 1G). In vivo, hMPLW515L adoptive transplant with Cxcr2KO hematopoietic donor cells demonstrated improved leukocytosis, thrombocytosis (Figure 2A) and splenomegaly in comparison to Cxcr2WT hMPLW515L recipient mice. Pathologic analysis revealed a reduction in reticulin fibrosis in bone marrow (Figure 2B) and spleen, translating into an improvement in overall survival (Figure 2C). Notably, a significant reduction in dysplastic MKs-a hallmark feature of MF-was also observed in Cxcr2KO hMPLW515L mice (Figure 2D) supporting a role for CXCR2 signaling in MK proliferation.
Conclusion: IL8 secreting clones are associated with increased symptom severity and fibrosis grade in MF. Gene expression of MF CD34+ IL8 secreting clones shows up-regulation of inflammatory genes S100A8/A9, implicated in myofibroblast proliferation. Cxcr2 KO abrogates fibrosis formation and prolongs survival in the hMPLW515L model, and CXCR1/2 inhibition impairs colony forming capacity of MF CD34+ cells. These data suggest pharmacologic inhibition of this pathway should be investigated as potential therapy in MF and in PV/ET patients at high risk of fibrotic transformation.
Fan:IsoPlexis: Current Employment, Current equity holder in private company; Singleron Biotechnologies: Current Employment, Current equity holder in private company. Levine:Morphosys: Consultancy; Prelude Therapeutics: Research Funding; Qiagen: Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees; Gilead: Honoraria; Loxo: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy; Amgen: Honoraria; Astellas: Consultancy; Imago: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; C4 Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Isoplexis: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Honoraria, Research Funding; Roche: Consultancy, Honoraria, Research Funding; Lilly: Consultancy, Honoraria; Janssen: Consultancy. Hoffman:Protagonist: Consultancy; Abbvie: Membership on an entity's Board of Directors or advisory committees; Dompe: Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees; Forbius: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal